
Information also can be obtained from scientific societies or associations such as the American Association of Pharmaceutical Scientists (AAPS) (7), the Parenteral Drug Association (PDA) (8), the Drug Information Association (DIA) (9), and the International Society of Pharmaceutical Engineering (ISPE) (10). There are many sources of advice on computerized system validation and analytical instrument qualification for the laboratory, including regulatory agencies, such as the United States Food and Drug Administration (FDA) (1,2) regulatory associations such as the Pharmaceutical Inspection Convention/Co-operation Scheme (PIC/S) (3,4) the Official Medicines Control Laboratories (OMCL) in Europe (5) and pharmacopeias such as the United States Pharmacopeia (USP) (6). This column explains the discrepancies between the two documents as well as changes now being made to both in an attempt to enable an integrated approach to qualification and validation of laboratory instruments and systems. 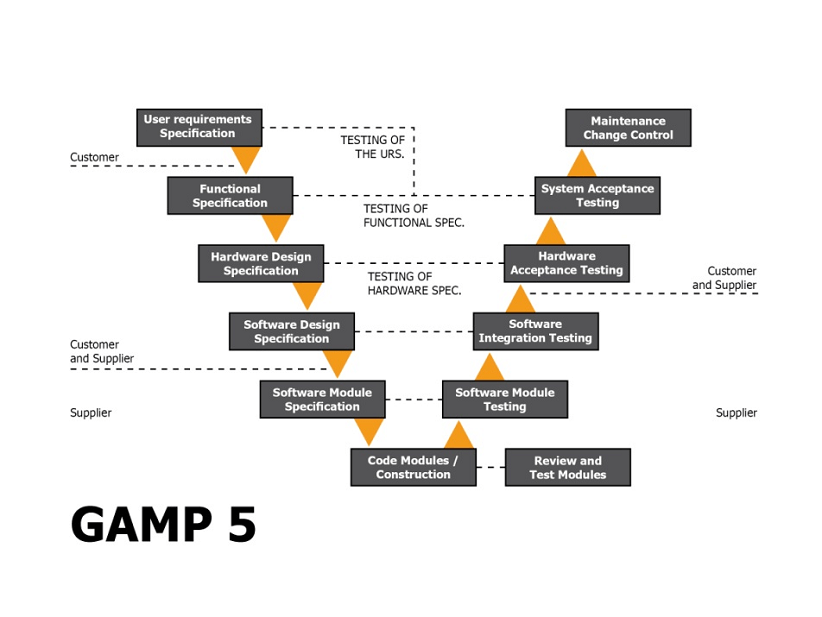
The United States Pharmacopeia general chapter on analytical instrument qualification ( USP ) and the ISPE's Good Automated Manufacturing Practice (GAMP) Good Practice Guide on laboratory computerized systems are the two main sources of guidance for qualifying analytical instruments and validating computerized systems used in the laboratory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
